 
Inner transition metals have a wide range of uses due to their unique properties. Metallic luster: Inner transition metals are typically shiny and have a metallic luster due to their high reflectivity of light.Similar chemical properties: The lanthanides and actinides have similar chemical properties, which makes them difficult to separate from each other.Oxidation states: Inner transition metals exhibit a range of oxidation states, including high oxidation states, due to the availability of multiple valence electrons in the f-orbitals.Radioactivity: Many inner transition metals are radioactive due to the presence of unstable isotopes in their nuclei.Complex ion formation: Inner transition metals can form complex ions due to the availability of empty f-orbitals, which can accommodate electrons from other atoms or molecules.Paramagnetism: Inner transition metals are paramagnetic, meaning that they are attracted to a magnetic field due to the presence of unpaired electrons in the f-orbitals.High melting and boiling points: Inner transition metals have high melting and boiling points due to the strong metallic bonding resulting from the presence of unpaired electrons in the f-orbitals.Electronic configuration: Inner transition metals have partially filled f-orbitals, which gives them unique electronic configurations and unusual chemical and physical properties.Here are some properties of inner transition metals: However, they were not widely studied or understood until the 20 th century. Inner transition metals have been known since ancient times, and were used for jewelry and decoration.However, they also pose environmental and health risks due to their radioactivity. The actinides are all radioactive, and some, like uranium and plutonium, are important for nuclear energy and weapons.However, they are not actually rare, and are found in many minerals. The lanthanides are also known as the “rare earth elements” because they are relatively scarce in the earth’s crust.For example, neodymium and samarium are used to make powerful magnets, and cerium is used in catalytic converters to reduce emissions from vehicles. Inner transition metals are important in technology and industry.Most inner transition metals are radioactive, and some have been used in nuclear reactors and weapons. 
This results in unusual chemical and physical properties, such as high melting and boiling points, complex ion formation, and paramagnetism. 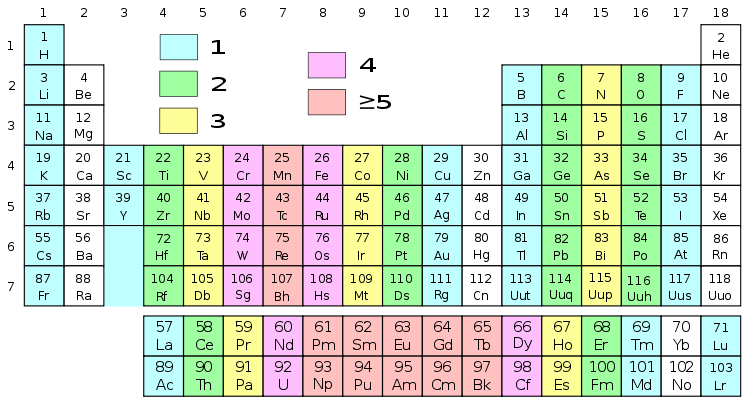
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
